General Information
| Immunogen: | synthetic peptides specific to XveloFL (H2N-QTREPRYETSKQGKQC-CONH2 and H2N-CKKNPKKSQSLSEPED-CONH2) and XveloSV (H2N-CSSLLKENKLELIQKK-COOH and H2N-CKIQPKSKSKISLQKR-CONH2). |
| Form: | polyclonal |
| ISOType: | |
| Animal: | Goat |
| Applications: | Wholemount, Section |
Attribution
| Origin: | Hugh Woodland, Warwick, UK |
| Literature: | Nijjar, S. and Woodland, H.R. "Protein interactions in Xenopus germ plasm RNP particles." PLoS One. 2013 Nov 12;8(11). http://www.ncbi.nlm.nih.gov/pubmed/24265795 |
Goat antibodies against unique regions of both XveloFL (long isoform) and XveloSV (short isoform).
“These antibodies proved ineffective in western blots of endogenous proteins, although they did detect over-expressed fusion proteins. The antibodies did work well in immunofluorescence staining of oocytes. We find that both isoforms of the endogenously expressed Xvelo protein are localised in the germ plasm RNPs of full-grown oocytes and fertilised eggs. The XveloFL is also localised to the Balbiani body of pre-vitellogenic oocytes.”
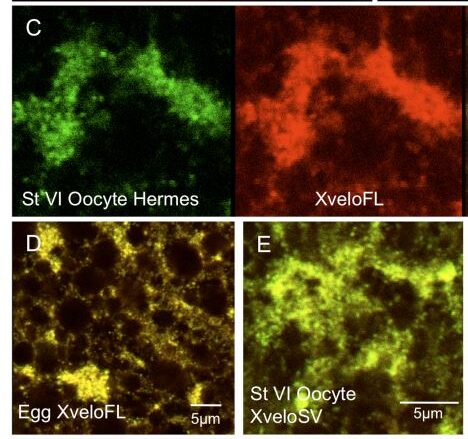
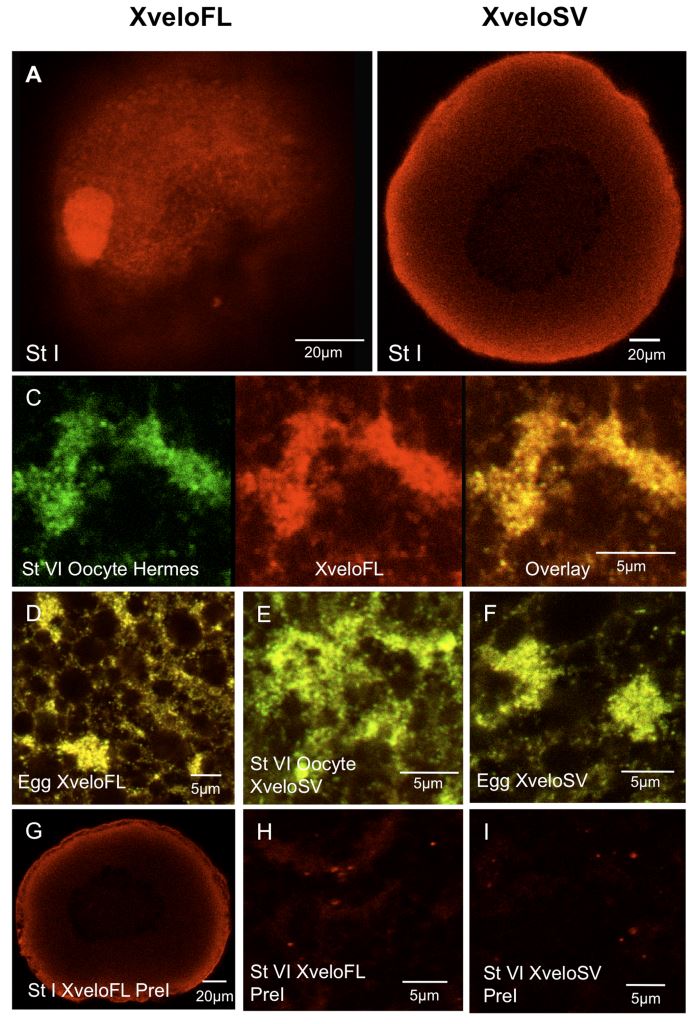
Figure 6. Expression of endogenous Xvelo isoforms during oogenesis. Oocytes, isolated with collagenase treatment to remove follicles, were fixed and stained with affinity-purified antibodies prior to confocal microscopy. (A–B) Previtellogenic oocytes stained for XveloFL and SV respectively. (C–F) The germ plasm region of stage VI oocytes and fertilized eggs. In (C) Hermes and XveloFL were stained and we show coincidence in an overlay in the right hand panel. In (D–F) only the overlays of Hermes (green) and Xvelo proteins (red) are shown. C) and (E) show stage VI oocytes and (D) and (F) show fertilized eggs. Alexa Fluor 488 and Dylight 594 conjugates were used as secondary antibodies for Hermes and Xvelo protein-stained oocytes respectively. Pre-immune serum for both XveloFL and SV is shown in panels G–H. The data was identical when secondary antibodies alone were used (not shown).