Egg Extracts
Xenopus laevis egg extracts
Many complex biological processes and their regulation can be reconstituted in vitro using extracts made from Xenopus laevis eggs. They are widely used for studying the cell cycle, DNA replication and repair, spindle assembly and microtubule motor function.
To make these extracts more easily available, we now make two kinds of extracts.
- Metaphase-arrested extracts are prepared from laid eggs in metaphase II of meiosis (often termed ‘CSF’ or cytostatic factor extracts), using Andrew Murray’s protocol (Murray, 1991) with minor modifications.
- Interphase extracts are made from eggs that have been briefly treated with calcium ionophore, since a rise in calcium triggers the destruction of CSF which leads to eggs entering interphase within a few minutes.
We snap-freeze the extracts and store them in liquid nitrogen until despatch on dry ice. Upon receipt they should be stored at -80°C. Frozen extracts are suitable for most studies, except spindle assembly.
For metaphase CSF extracts, we offer two grades. Grade I extracts hold their metaphase arrest when thawed and incubated at room temperature, and will also enter interphase when calcium is added. An example is shown in the figure. Grade II metaphase extracts hold their metaphase arrest when kept on ice, but either come out of arrest at room temperature, or do not activate fully when calcium is added. These extracts are suitable for many biochemical experiments.
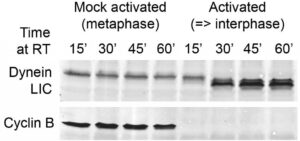 Figure 1. Example of a meiotic metaphase II-arrested CSF extract. The extract retained its metaphase arrest after incubation at room temperature without calcium addition, while calcium addition triggered entry into interphase. Cyclin B is stable in the CSF extract but degraded within 15 mins of calcium addition. Dynein LIC becomes dephosphorylated after entry into interphase (Niclas et al., 1996; Addinall et al., 2001).
Figure 1. Example of a meiotic metaphase II-arrested CSF extract. The extract retained its metaphase arrest after incubation at room temperature without calcium addition, while calcium addition triggered entry into interphase. Cyclin B is stable in the CSF extract but degraded within 15 mins of calcium addition. Dynein LIC becomes dephosphorylated after entry into interphase (Niclas et al., 1996; Addinall et al., 2001).
References:
Murray, A (1991). Cell cycle extracts. Meths. Cell Biol. 36:581-605







